 The volume is definite (does not change) if the temperature and pressure are constant. LiquidsĪ liquid is a fluid that conforms to the shape of its container but that retains a nearly constant volume independent of pressure. A solid can also change directly into a gas through a process called sublimation. Ice has 15 known crystal structures, each of which exists at a different temperature and pressure.Ī solid can transform into a liquid through melting, and a liquid can transform into a solid through freezing. For example, iron has a body-centered cubic structure at temperatures below 912 ☌ and a face-centered cubic structure between 912 and 1,394 ☌. There are many different crystal structures, and the same substance can have more than one structure. In crystalline solids, particles are packed in a regularly ordered, repeating pattern. Solids can only change shape under force, as when broken or cut. As a result, a solid has a stable, definite shape and a definite volume. The forces between the particles are strong enough that the particles cannot move freely they can only vibrate. SolidsĪ solid’s particles are packed closely together. This diagram shows the nomenclature for the different phase transitions. Each of these three classical states of matter can transition directly into either of the other two classical states. Solid is the state in which matter maintains a fixed volume and shape, liquid is the state in which matter adapts to the shape of its container but varies only slightly in volume, and gas is the state in which matter expands to occupy the volume and shape of its container. Historically, the states of matter were distinguished based on qualitative differences in their bulk properties. Much of the atomic matter of the universe is hot plasma in the form of rarefied interstellar medium and dense stars. Further states, such as quark-gluon plasmas, are also believed to be possible. In extreme environments, other states may be present, such as plasma, Bose-Einstein condensates, and neutron stars. The three states of matter are the three distinct physical forms that matter can take in most environments: solid, liquid, and gas. solid: A substance that retains its size and shape without a container a substance whose molecules cannot move freely except to vibrate.gas: A substance that can only be contained if it is fully surrounded by a container (or held together by gravitational pull) a substance whose molecules have negligible intermolecular interactions and can move freely.It takes the shape of its container but maintains constant volume. liquid: A substance that flows and keeps no definite shape because its molecules are loosely packed and constantly moving.Gaseous matter is composed of particles packed so loosely that it has neither a defined shape nor a defined volume.Particles can move about within a liquid, but they are packed densely enough that volume is maintained. Liquid matter is made of more loosely packed particles.A solid will retain its shape the particles are not free to move around. Solid matter is composed of tightly packed particles.Matter can exist in one of three main states: solid, liquid, or gas.It is the only metal we know of that is liquid at room temperature. Particles are free to move over each other, but are still attracted to each otherĪ familiar liquid is mercury metal. 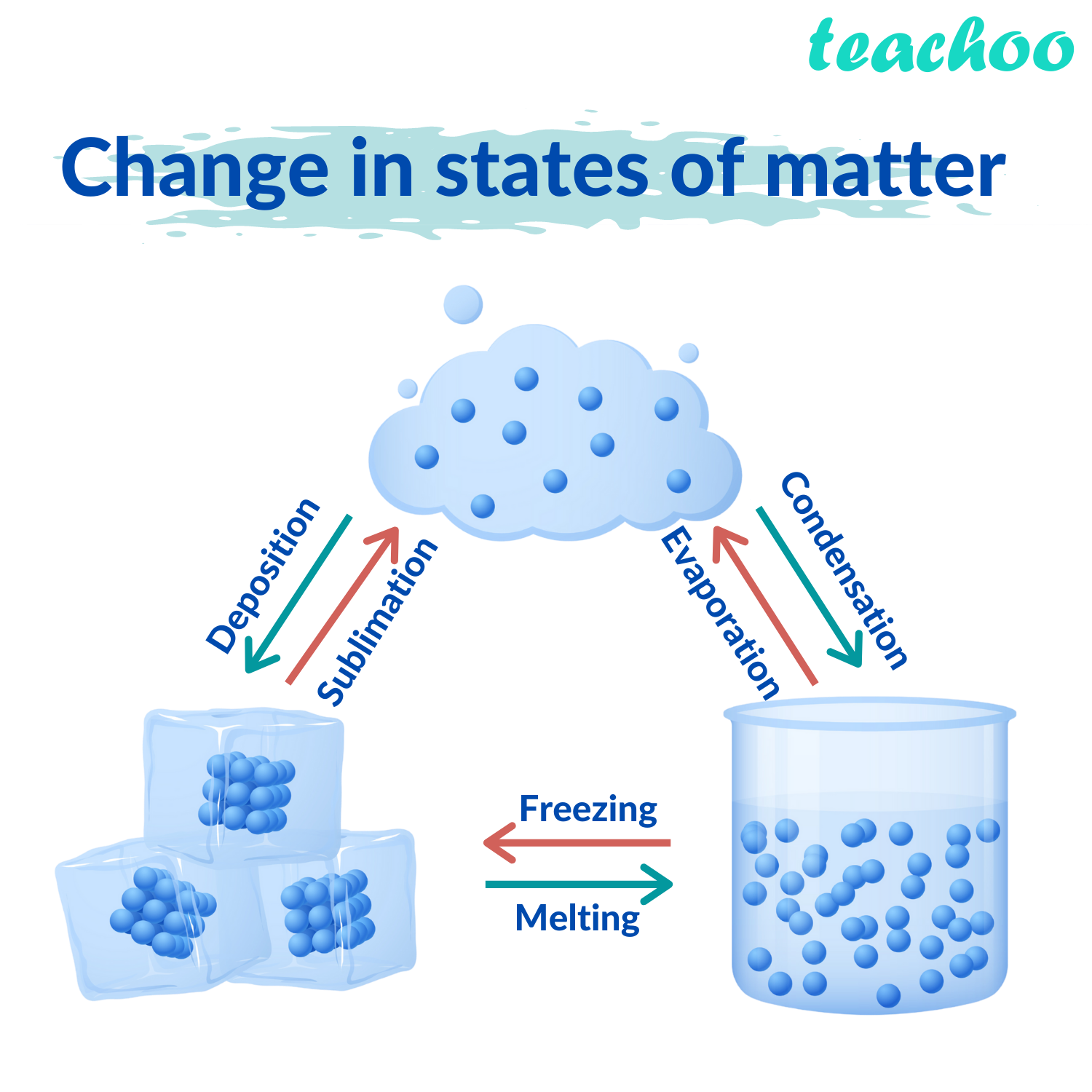
No definite shape (takes the shape of its container).Liquids have the following characteristics: However, because the particles can move about each other rather freely, a liquid has no definite shape and takes a shape dictated by its container. In a liquid, the particles are still in close contact, so liquids have a definite volume. If the particles of a substance have enough energy to partially overcome intermolecular interactions, then the particles can move about each other while remaining in contact. Images used with permission (public domain) Note that, as in the crystal, each Silicon atom is bonded to 4 oxygen atoms, where the fourth oxygen atom is obscured from view in this plane. 
(right) The random network structure of glassy \(SiO_2\) in two-dimensions. \): (left) The periodic crystalline lattice structure of quartz \(SiO_2\) in two-dimensions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
